A | B | C | D | E | F | G | H | CH | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9
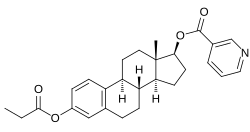
Estrapronicate
 | |
| Clinical data | |
|---|---|
| Other names | Estradiol propionicotinate; Estradiol propionate nicotinate; Estradiol nicotinate propionate; Estradiol 17β-nicotinate 3-propionate |
| Routes of administration | Intramuscular injection |
| Drug class | Estrogen; Estrogen ester |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.021.786 |
| Chemical and physical data | |
| Formula | C27H31NO4 |
| Molar mass | 433.548 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Estrapronicate (INN), also known as estradiol nicotinate propionate is an estrogen medication and estrogen ester which was never marketed.[1] It was studied as a component of the experimental tristeroid combination drug Trophobolene, which contained nandrolone decanoate, estrapronicate, and hydroxyprogesterone heptanoate.[2][3]
See alsoedit
- List of estrogen esters § Estradiol esters
- Estrapronicate/hydroxyprogesterone heptanoate/nandrolone undecanoate
Referencesedit
- ^ Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 898–. ISBN 978-1-4757-2085-3.
- ^ Excerpta medica. Section 8, Neurology and neurosurgery. 1981. p. 10.
- ^ Testosterone Congeners—Advances in Research and Application: 2013 Edition: ScholarlyBrief. ScholarlyEditions. 21 June 2013. pp. 137–. ISBN 978-1-4816-9288-5.
>Text je dostupný pod licencí Creative Commons Uveďte autora – Zachovejte licenci, případně za dalších podmínek. Podrobnosti naleznete na stránce Podmínky užití.
Zdroj: Wikipedia.org - čítajte viac o Estrapronicate
Text je dostupný za podmienok Creative
Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších
podmienok.
Podrobnejšie informácie nájdete na stránke Podmienky
použitia.

